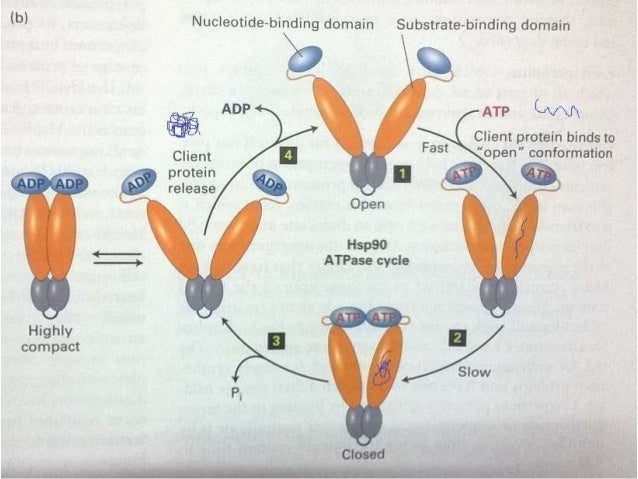
35 To date, no clear molecular function for myocilin has been established, 10, 36 and myocilin double knockout or overexpressing mice show no clear phenotype. 14, 27 – 33 Myocilin interactions have also been demonstrated with a number of molecules associated with cell signaling and metabolism, including glyceraldehyde-3-phosphate dehydrogenase (GAPDH), 34 and optimedin, which is itself an olfactomedin-related protein. Myocilin associations have been reported with mitochondria, 22 – 24 exosome-like vesicles, 25, 26 and components of the cytoskeleton and extracellular matrix. 8, 10 On gels, the primary band runs as a doublet of approximately 55 and 57 kDa, with a 66-kDa band apparent in some cases. 8, 10, 17 – 21 Both intracellular nonglycosylated and extracellular glycosylated and nonglycosylated forms of myocilin have been identified. 8 Myocilin is widely distributed in ocular tissues, 10, 12 – 16 and its mRNA is also expressed in various nonocular tissues, including the heart, skeletal muscle, kidney, and peripheral nerves. 8, 10, 11 It includes structural motifs for both N- and O-linked glycosylation, hyaluronan binding, and a myosin-like leucine zipper. 9 MYOC, the myocilin gene, codes for a 504-aa glycoprotein with homology to olfactomedin, a mucoid protein found in neuroepithelium. 7, 8 identified and characterized a TM-inducible glucocorticoid response protein, which is also known as myocilin. 2 Myocilin mutations have been associated with juvenile and early onset POAG and a subset of adult POAG. 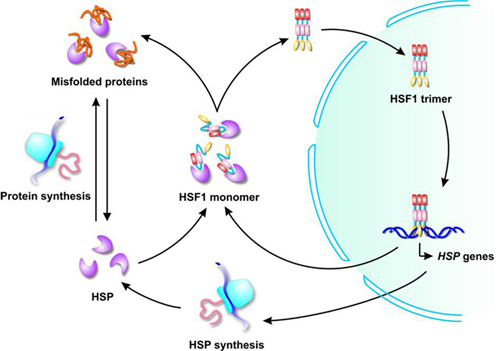
1 The most common form, primary open-angle glaucoma (POAG), is typically associated with elevated intraocular pressure, loss of peripheral vision, and accompanying damage to the optic nerve. Glaucoma, which affects approximately 70 million people, is a major cause of blindness throughout the world. One function of myocilin may be to serve as a molecular chaperone. Myocilin also suppresses the thermal aggregation of citrate synthase. Myocilin is induced in response to several cellular stresses and displays general molecular chaperone activity by protecting DrdI, citrate synthase, and GAPDH from thermal inactivation. Myocilin at 18 nM was more effective than 1 μM bovine serum albumin at protecting DrdI from thermal inactivation. Myocilin also protected GAPDH from thermal inactivation for 10 minutes at 45☌. Myocilin significantly reduced thermal aggregation of citrate synthase to levels 36% to 44% of control levels. Myocilin protected citrate synthase activity against thermal inactivation for 5 minutes at 55☌ in a concentration-dependent manner, with nearly full protection of 1.5 μM citrate synthase in the presence of 650 nM myocilin. Levels of extracellular myocilin expressed by TM cells were increased in response to mechanical stretch, heat shock, TNFα, or IL-1α. Myocilin induction was assessed after exposure of TM cells to several types of stress treatments. Light scattering was used to evaluate thermally induced aggregation of citrate synthase. Its ability to protect citrate synthase, glyceraldehyde-3-phosphate dehydrogenase (GAPDH), and the restriction endonuclease DrdI from thermal inactivation was evaluated. Myocilin was isolated and purified from porcine trabecular meshwork (TM) cell culture media. Studies were conducted to see whether myocilin can act as a general molecular chaperone. Myocilin is thought to be a stress response protein, but its exact molecular functions have not been established.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
